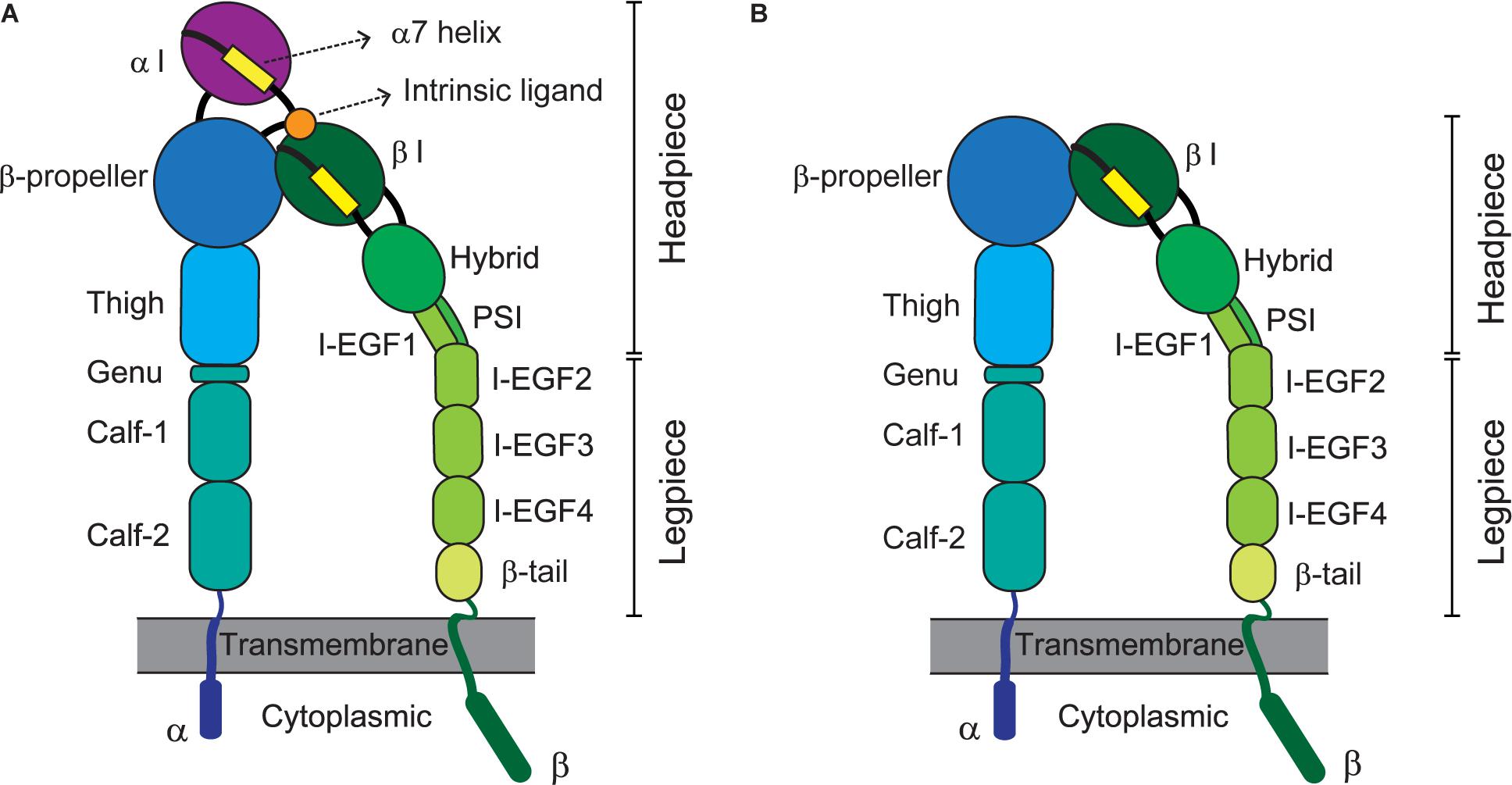
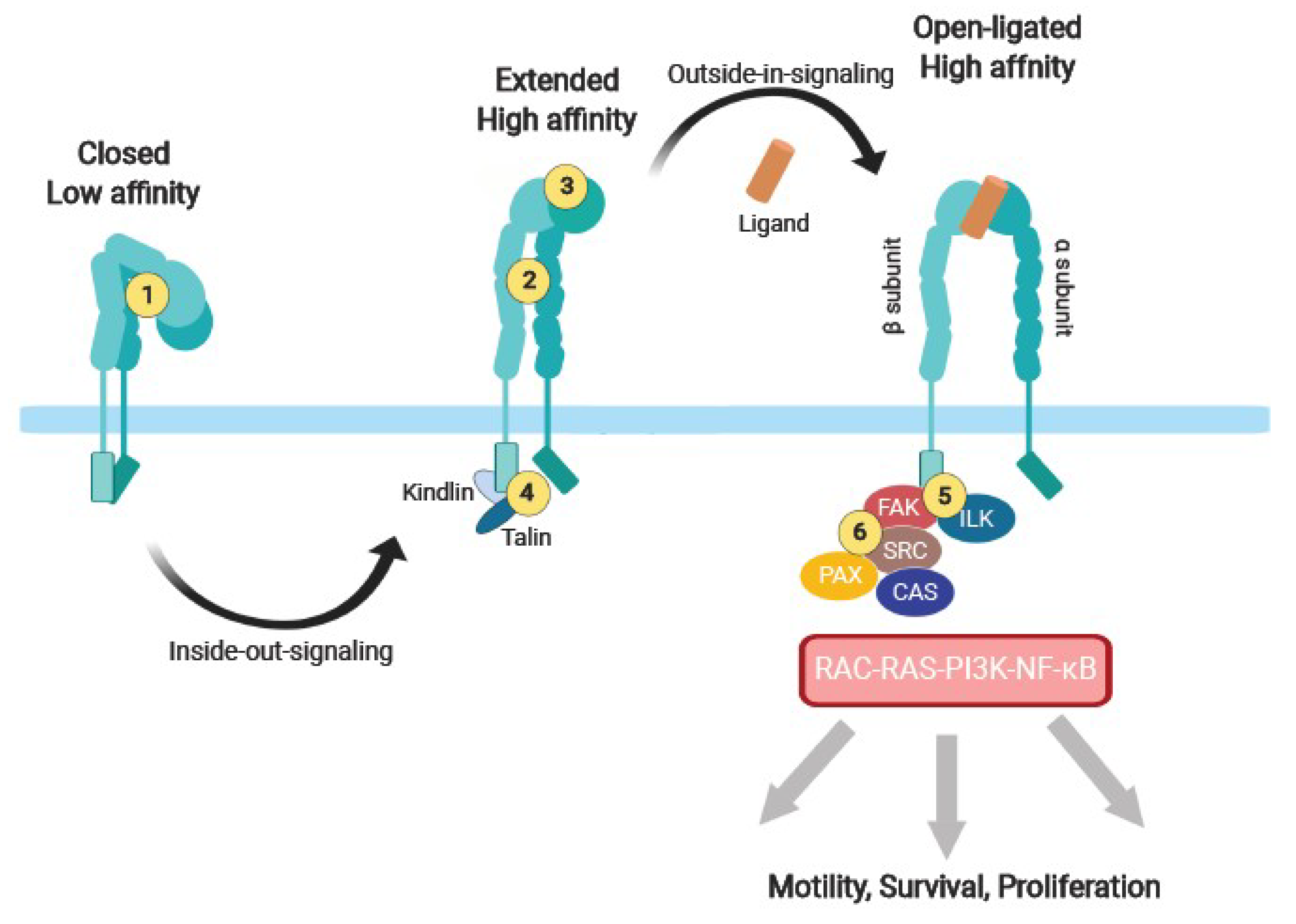
In this context, integrins αvβ6 and αvβ8 regulate the release of TGF-β from its latency-associated protein and its ability to interact with TβRs on nearby cells ( 11– 13). Integrins also regulate matrix homeostasis by modulating matrix expression and degradation, altering the activation of specific receptor tyrosine kinases, or controlling the activation and levels of growth factors like TGF-β ( 10). Upon ligand binding, integrins initiate multiple intracellular signaling pathways that regulate critical cellular functions such as migration, survival, and proliferation ( 9). In mammalian cells, integrins combine to form 24 different heterodimers with different ligand specificity to matrix molecules ( 8). They are transmembrane receptors for extracellular matrix components formed by 2 noncovalently associated α and β subunits. Integrins are also potent regulators of matrix homeostasis ( 1, 2). Whether the remaining tyrosine residues are also involved in signaling and control TβRII-mediated SMAD activation and fibrotic signaling is currently unknown. TβRIIY284 was shown to be phosphorylated by Src and implicated in TβR-mediated noncanonical p38 MAPK activation ( 7). The TβRII cytoplasmic domain also contains 5 phosphorylatable tyrosines: Y259, Y284, Y336, Y424, and Y470 ( 6, 7). Signaling from TβRII to TβRI is primarily modulated by the autophosphorylation of 3 serine residues in the TβRII cytoplasmic tail: S213 and S409 promote kinase activity and interaction with TβRI, while S416 inhibits the receptor ( 5). The activated TβRI, in turn, promotes serine phosphorylation of SMAD2 and SMAD3, their association with SMAD4, translocation to the nucleus, and transcription of profibrotic genes ( 3, 4). TGF-β exerts its functions by binding of the constitutively active type II TGF-β receptor (TβRII), which leads to serine phosphorylation and activation of TβRI (also known as ALK5). The cytokine TGF-β is one of the most potent stimulators of fibrosis following chronic injury. Growth factors and cytokines are important regulators of matrix homeostasis ( 1, 2). Thus, there is great interest in deciphering the molecular mechanisms controlling matrix homeostasis in normal and pathological states in order to devise effective therapies. Although fibrosis is promoted by a range of different factors, including genetic predisposition, cytokines, matrix receptors, and oxidative stress ( 1), an accepted treatment is still not available. Fibrosis is irreversible and disrupts the normal tissue architecture, eventually leading to organ dysfunction and failure. Introductionįibrosis is characterized by the uncontrolled deposition of extracellular matrix components after tissue injury and is the hallmark of many chronic diseases. Together, these findings uncover a crosstalk between integrin α1β1 and TβRII that is essential for TβRII-mediated SMAD activation and fibrotic signaling pathways. The collagen-binding receptor integrin α1β1 was required for recruitment of TCPTP to the TβRII tail, as mice lacking this integrin exhibited impaired TCPTP-mediated tyrosine dephosphorylation of TβRII that led to severe fibrosis in a unilateral ureteral obstruction model of renal fibrosis.
Conversely, the T cell protein tyrosine phosphatase (TCPTP) dephosphorylated TβRII tail tyrosine residues, resulting in inhibition of TβR-dependent fibrotic signaling. Here, we determined that phosphorylation of tyrosines within the TβRII tail was essential for SMAD-dependent fibrotic signaling within cells of the kidney collecting duct. Autophosphorylation of serine residues within TβRII is considered the principal regulatory mechanism of TβRII-induced signaling however, there are 5 tyrosine residues within the cytoplasmic tail that could potentially mediate TβRII-dependent SMAD activation.
TGF-β mediates both the development and the progression of kidney fibrosis through binding and activation of the serine/threonine kinase type II TGF-β receptor (TβRII), which in turn promotes a TβRI-mediated SMAD-dependent fibrotic signaling cascade. Tubulointerstitial fibrosis underlies all forms of end-stage kidney disease.
